Project Description
Around 20% of people who suffer a heart attack will progress to heart failure within five years due to the damage inflicted to the cardiac tissue. The cardiac regeneration therapy project was focused on preventing the onset of heart failure by further developing an injectable hydrogel with Dr. Kevin Healy, Department of Bioengineering Chair at UC Berkeley, and Dr. Mark Ratcliffe, Professor of Surgery at UCSF. The hydrogel is designed to be injected into the wall of the heart at the area of injury. When raised to body temperature, the thermoresponsive material in the hydrogel stiffens and provides non-contractile mechanical strength to the damaged region.
The framework for the project team was divided into four main areas: addressing fundamental biocompatibility concerns, performing material characterization, refining a computational model of the heart/material interactions, and developing a Food and Drug Agency approval plan for this material as a medical device.


The experience of working as a part of a team with three other students, all with different backgrounds, taught me the importance of collaboration, and how looking at things from different angles can be very rewarding and usually yields the most creative outcomes. Working in a team also improved my communication skills and taught me invaluable skills for entering into engineering management, and I am very thankful to the Fung Institute for endowing me with that experience. Additionally, before entering into the program, I felt that my presentation skills were weak, yet during this 10 month rigorous curriculum (both via the Fung Institute and the Bioengineering Department) which required frequent high-caliber presentations, I feel as though my confidence to deliver a compelling presentation has really increased.
— Amy Foley, BioE
Project Objective
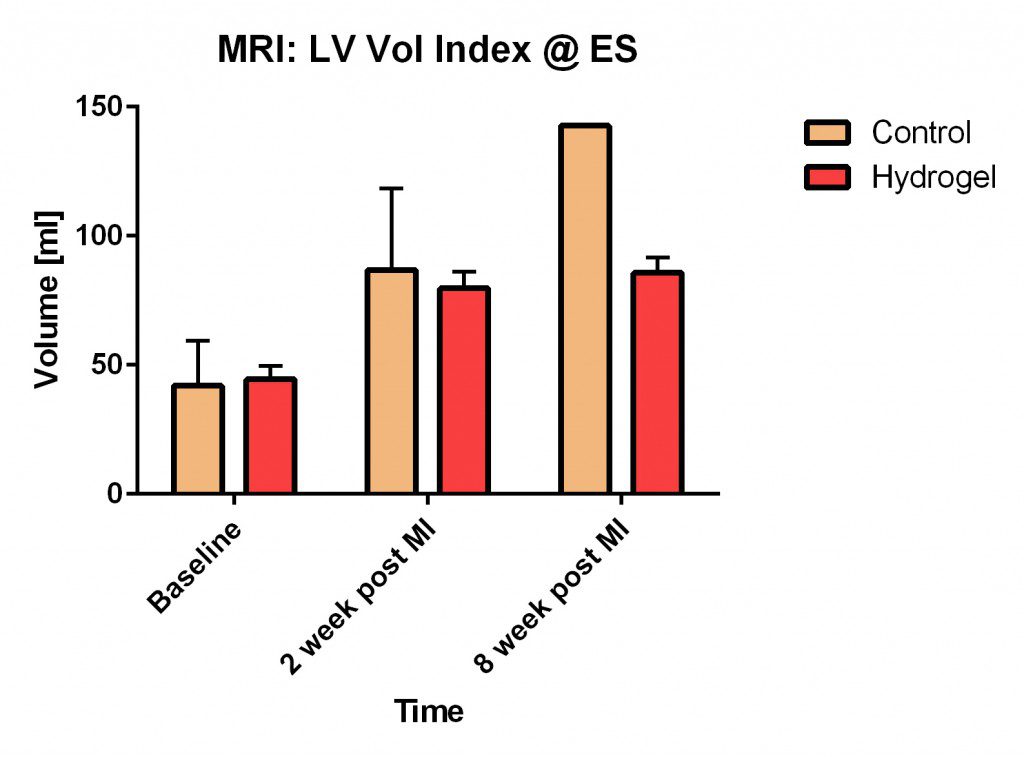
The graph shows two control sheep (induced myocardial infarction only, no treatment) and two sheep treated with the hydrogel injection. The plot shows the volume of the left ventricle over time. Shortly after a heart attack, the infarct and surrounding borderzone area thins and expands, which leads to an overall dilation of the left ventricle; this represents a significant step towards heart failure. The average left ventricular volume of the treated animals is less than the untreated, control animals, suggesting that the injection of the hydrogel material into the myocardium attenuated the adverse cardiac remodeling typically seen after a heart attack.
Value Created
There are approximately 720,000 Americans that have a heart attack every year. The direct and indirect costs for treating and sustaining heart failure patients in 2009 was estimated at $45B, and is expected to increase to $220B by 2030 (Go et al. 2013:e109/e114). Furthermore, by 2030 it is forecasted that every US taxpayer will pay $244 a year to care for heart failure patients alone (Mintel 2013). This hydrogel has the therapeutic potential to prevent incidences of heart failure through immediate treatment following an initial heart attack. Even if the total heart failure population was only marginally reduced, the economic impact of the hydrogel product would be immense.


Accomplishments
During the later part of the Spring 2015 semester, the team worked with Dr. Ratcliffe and his Cardiac Biomechanics Laboratory team to conduct ovine animal studies. Preliminary results with this small animal trial (n=2) show that the hydrogel material reduced end-diastolic volume of the left ventricle more so relative to a control animal at the 2 and 8 week time points after a heart attack. The archetypal ventricular response after injury is an expansion of the infarct area and ventricular dilation (Jugdutt 2003). Therefore, the reduction of left ventricular volume relative to the control indicates that the hydrogel minimized ventricular expansion and interfered with the conventional remodeling process.

This project introduced me to new realms of engineering and made me appreciate the complexities of developing a brand new technology from the very early phases of research. In parallel, our papers and numerous presentations outside of the lab really helped to enhance our communication skills, nicely complementing our technical skill sets. It also provided a great opportunity to learn from all of my teammates since the 4 of us came from completely different backgrounds.
— Minori Keefe, BioE

Faculty Advisor
Kevin Healy: Department Chair, Department of Bioengineering.
Together with Dr. Mark Ratcliffe, my laboratory has pioneered bioinspired hydrogels for matrix-assisted infarct stabilization as a treatment for heart attack patients. We believe this therapy has great potential to ameliorate the negative effects on cardiac tissue and heart function after a heart attack. The students that worked on this master of engineering project were critical in moving this technology closer to a clinical reality.
Sources
- Go, A. S., et al. 2013 Heart Disease and Stroke Statistics – 2013 Update. Circulation. 127:e6-e245.
- Jugdutt, B. I. 2003 Ventricular Remodeling After Infarction and the Extracellular Collagen Matrix: When Is Enough Enough? Circulation 108(11): 1395-403.
- Mintel: Global Market Research & Market Insight. 2013 Study: Heart failure costs could double by 2030. www.mintel.com, accessed February 16, 2015.
.
← View all Capstone Projects